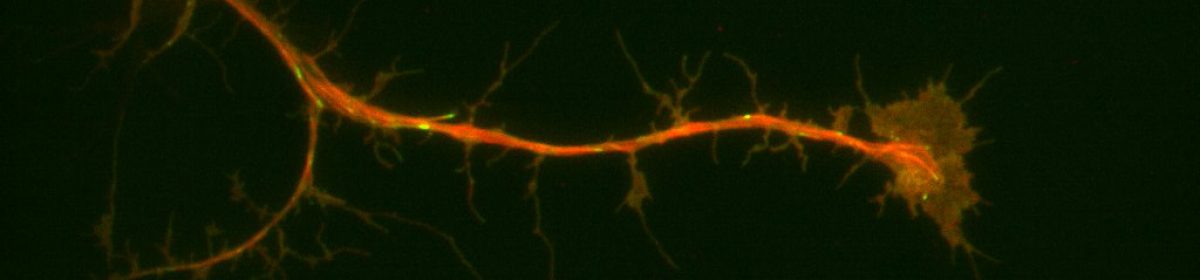
Studies have shown that the cytoskeletal elements in the cell (actin, microtubules and intermediate filaments) engage in extensive crosstalk. This crosstalk is an important part of the regulation of the cytoskeleton, as well as a number of other biological processes. For this blog post, I will be focusing on actin-microtubule cross linking, since that is most relevant to my thesis research. Before I get into specifics about how this is related to my thesis, I’m going to give a brief, general overview of actin microtubule crosstalk and the various roles it can play within a cell.
Actin is a highly conserved protein in cells that switches between G-actin and filamentous F-actin. Actin is one of the most abundant proteins in eukaryotic cells and plays an important role in muscle contraction, cell signalling and regulation of cell shape. Microtubules are dynamic, polar and are made up of alpha & beta tubulin. The tubulin polymerizes to form microtubule filaments. Key to microtubules ability to perform their functions in the cell is dynamic instability and polarity. Dynamic instability is characterized by periods of rapid growth followed by periods of depolymerization. This allows microtubules to rapidly alter their configuration in order to fit the needs of the cell. Microtubules and actin play important roles in cell division, cell migration and other important cellular processes.
Actin Microtubule crosstalk is mainly defined through the physical mechanisms by which it occurs. This means that usually what happens is a physical linkage between parts of actin and parts of microtubules that lead to stabilization or nucleation etc. One of the main forms of actin microtubule crosstalk is crosslinking. This occurs when proteins link microtubules to actin. This linkage is enabled by large protein complexes that can also interact with microtubule plus end binding proteins. This linkage connects the plus ends of microtubules to actin bundles which can result in a redirection of microtubule growth.
One of the proteins that plays a role in actin microtubule cross linking is Short Stop (Shot). Short stop is a spectraplakin that has been shown to bind microtubules & actin filaments and has also been localized in growth cones. In my thesis, I’m looking at microtubules in Drosophila melanogaster neuroblasts, specifically at what happens when you knock down shot thus inhibiting crosslinking. I’m studying the effect of knocking down this crosslinking protein on microtubule dynamics in 3 different parts of the neuron.
Works Cited
Applewhite, D.A., Grode, K.D., Keller, D., Zadeh, A.D., Slep, K.C., and Rogers, S.L. (2010). The Spectraplakin Short Stop Is an Actin–Microtubule Cross-Linker That Contributes to Organization of the Microtubule Network. Molecular Biology of the Cell 21, 1714–1724.
Dogterom, M., and Koenderink, G.H. (2018). Actin–microtubule crosstalk in cell biology. Nature Reviews Molecular Cell Biology.
Sanchez-Soriano, N., Travis, M., Dajas-Bailador, F., Goncalves-Pimentel, C., Whitmarsh, A.J., and Prokop, A. (2009). Mouse ACF7 and Drosophila Short stop modulate filopodia formation and microtubule organisation during neuronal growth. Journal of Cell Science 122, 2534–2542.